Specific heat capacity is a key concept in thermal physics that helps us understand how different materials respond to heat energy. It is especially important when studying temperature changes, energy transfer, and material properties.
Specific Heat Capacity Meaning
The specific heat capacity meaning goes beyond just temperature change—it reflects how resistant a substance is to heating. Materials with high values require more energy input, making them excellent for thermal stability.
This is why substances like metals heat up quickly, while liquids like water resist temperature changes.
Specific Heat Capacity Definition (Scientific Context)
The specific heat capacity definition in scientific terms includes both energy and molecular behavior:
It is the heat energy required to raise the temperature of 1 kg of a substance by 1°C, determined by how particles store and transfer energy internally.
This internal storage includes:
- Vibrational energy
- Rotational motion
- Intermolecular forces
Specific Heat Capacity Formula and Units
The specific heat capacity equation is:
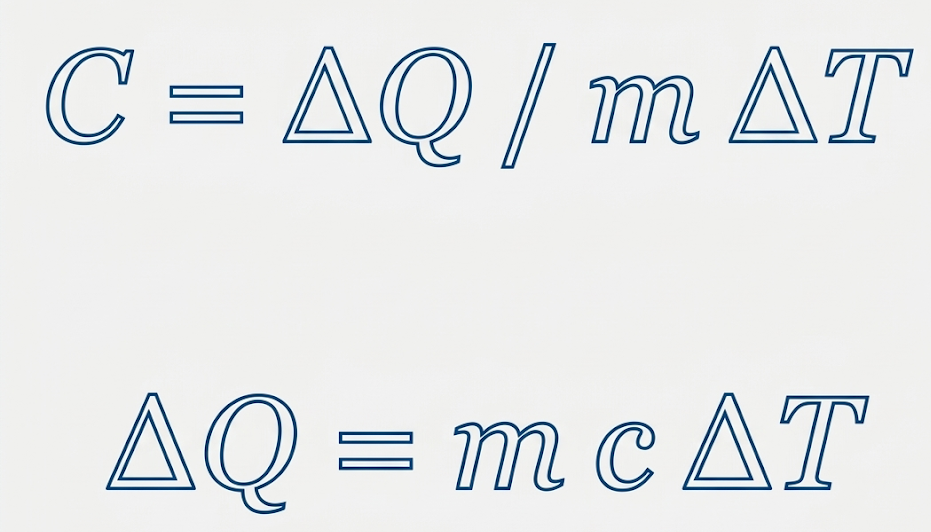
Specific heat capacity refers to the amount of heat energy needed to raise the temperature of 1 kilogram of a substance by 1 Kelvin (or 1°C).
Units of Specific Heat Capacity
Understanding units is essential, especially for solving numerical problems in exams like GCSE.
- In the SI system, the unit is:
J kg⁻¹ K⁻¹ (joules per kilogram per Kelvin) - In the CGS system, it is commonly expressed as:
cal/g°C or J/g°C
Dimensional Formula
The dimensional formula of specific heat capacity is:
[M⁰ L² T⁻² K⁻¹]
What is the Specific Heat Capacity of Water?
The specific heat capacity of water at normal temperature and pressure (NTP) is approximately 4.2 J/g°C, which is the same as 4,186 J/kg°C or 1 cal/g°C. This means that 4.2 joules of heat energy are required to raise the temperature of 1 gram of water by 1°C.
This standard value is often referred to in different ways, such as the heat capacity of water, specific heat (H₂O), or thermal energy capacity of water, but they all describe the same property.
Compared to most substances, this value is quite high. Water does not transfer heat easily, so it can absorb a large amount of energy before its temperature increases significantly.
Even in different states, water maintains relatively high values:
- Water (liquid): ~4.2 J/g°C
- Water vapor (gas): ~1.9 J/g°C
- Ice (solid): ~2.06 J/g°C
This shows that water retains its strong heat-storing ability across all phases.
Because of this property, water heats up and cools down slowly. It can absorb or release large amounts of heat without sudden temperature changes, allowing it to maintain a stable temperature for longer periods.
This characteristic is especially important in the human body. Since the body contains a large proportion of water, it helps maintain a stable internal temperature around 37°C, even when external conditions vary. If water had a lower heat capacity, the body would be far more sensitive to rapid temperature changes, increasing the risk of overheating or excessive cooling.
Why the Specific Heat Capacity of Water is Exceptionally High
The specific heat capacity of water is much higher than most liquids because of the way its molecules interact and store energy. Water molecules are connected by strong hydrogen bonds, and a large amount of heat is required to break or loosen these bonds before the temperature can rise.
In addition, water does not use incoming heat energy only to increase temperature. A significant portion of this energy goes into rearranging its molecular structure, which slows down the overall temperature increase.
Heat energy in water is also spread across different forms of internal motion, such as molecular movement and vibrations. Because energy is distributed in multiple ways, more heat is needed to produce a noticeable temperature change.
As a result, water can absorb and store large amounts of heat, which is why its thermal capacity is significantly higher than most other substances.
Specific Heat Capacity of Different Substances
The value of specific heat capacity varies depending on both the material and its physical state (solid, liquid, or gas). Substances like water generally have higher values, while metals tend to have lower values, meaning they heat up more quickly.
Below is a table showing typical values:
| Material | State | Specific Heat Capacity (J/g°C) |
| Water | Liquid | 4.18 |
| Water (Ice) | Solid | 2.06 |
| Water (Steam) | Gas | 1.87 |
| Ammonia | Gas | 2.09 |
| Ethanol | Liquid | 2.44 |
| Aluminum | Solid | 0.90 |
| Graphite (Carbon) | Solid | 0.71 |
| Copper | Solid | 0.39 |
| Gold | Solid | 0.13 |
| Iron | Solid | 0.45 |
| Lead | Solid | 0.13 |
| Mercury | Liquid | 0.14 |
| Silver | Solid | 0.23 |
Key Observations
- Water (liquid) has one of the highest values, showing its strong ability to store heat.
- Metals like copper, silver, and gold have low values, so they heat and cool quickly.
- The same substance (like water) shows different values in different states, highlighting the role of molecular structure.
Specific Heat at Constant Pressure and Constant Volume
In thermodynamics, specific heat capacity is often studied under two different conditions—constant pressure (Cp) and constant volume (Cv). These concepts are especially important when dealing with gases, engines, and energy systems.
Specific Heat at Constant Pressure (Cp)
The specific heat at constant pressure (Cp) refers to the amount of heat energy required to raise the temperature of a substance by 1°C (or 1 K) while the pressure remains constant.
Under this condition, the substance is allowed to expand when heated. As a result, the heat supplied is used for two purposes:
- Increasing the temperature of the substance
- Doing external work during expansion
Because of this extra work, Cp is always greater than Cv.
Example:
For air at room temperature:
- Cp ≈ 1005 J/kg·K
This value is widely used in atmospheric science, engineering, and HVAC systems.
Specific Heat at Constant Volume (Cv)
The specific heat at constant volume (Cv) is the heat required to raise the temperature of a substance by 1°C (or 1 K) while keeping its volume fixed.
Since the volume does not change:
- No expansion takes place
- No external work is done
Therefore, all the heat energy goes directly into increasing the internal energy (temperature) of the substance.
Example:
For air at room temperature:
- Cv ≈ 718 J/kg·K
Key Relationship Between Cp and Cv
For gases, the relationship between Cp and Cv is:
- Cp – Cv =n R
where
- CV is heat capacity at constant volume
- CP is heat capacity at constant pressure
- R is the molar gas constant
- n is amount of substance
The value of the Gas Constant, R = 8.3145 J mol-1 K-1
Heat Capacity Ratio (Cp/Cv)
In thermodynamics, the heat capacity ratio is defined as the ratio of the specific heat at constant pressure (Cp) to the specific heat at constant volume (Cv). It is an important property used to describe the behavior of gases during different thermodynamic processes.
This ratio is commonly known as the adiabatic index or isentropic expansion factor, and it is represented by the symbol γ (gamma).
For an ideal gas, the relationship is:
γ = Cp / Cv
Since Cp includes both temperature change and expansion work, while Cv only accounts for temperature change, the value of γ is always greater than 1.
Typical Values of γ
- Air: ~1.4
- Monatomic gases (e.g., helium): ~1.67
- Diatomic gases (e.g., oxygen, nitrogen): ~1.4
Why is Cp Greater than Cv?
The specific heats of a gas are represented by Cp (at constant pressure) and Cv (at constant volume). Both indicate the amount of heat needed to raise the temperature of a unit mass by 1°C (or 1 K), but the conditions under which heating occurs make the difference.
According to the first law of thermodynamics:
ΔQ=ΔU+ΔW
Where:
- ΔQ = heat supplied to the system
- ΔU = change in internal energy
- ΔW = work done by the system
When a gas is heated at constant pressure, it is free to expand. So, the heat supplied is used for:
- Increasing the internal energy (ΔU)
- Doing external work (ΔW) during expansion
However, at constant volume, the gas cannot expand:
- No work is done (ΔW = 0)
- All the heat goes only into increasing internal energy (ΔU)
Because extra energy is required to perform work at constant pressure, more heat is needed to raise the temperature by the same amount.
Therefore, Cp is always greater than Cv (Cp > Cv).
Solved Examples on Specific Heat Capacity
Example 1: Calculate the heat required to raise 1 kg of sand from 20°C to 80°C.
(Specific heat of sand = 830 J/kg°C)
Given Data:
- m=1kg
- c=830J/kg°C
- ΔT=80−20=60°C
Q=mcΔT
=1×830×60
Q=49,800J
Example 2-Determine the temperature change if 25 kg of water absorbs 210 kJ of heat.
Given data:
Where:
- Q=210kJ=210,000J
- m=25kg
- c=4186J/kg°C (for water)
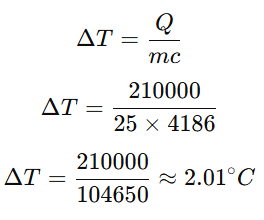
The temperature increase is approximately 2°C
Uses of Specific Heat Capacity
The concept of specific heat capacity is very useful in daily life. It helps us understand why some materials heat up quickly while others take longer.
1. Building Insulation
Materials with high heat capacity, like wood, are used in construction.
- Wood has a value of about 1,700 J/kg°C, which means it does not heat up or cool down quickly.
- This helps keep houses warm in winter and cool in summer.
That’s why wooden houses are suitable for areas with very hot or very cold climates.
2. Water in Swimming Pools
Water has a very high value (about 4,186 J/kg°C).
- Because of this, it heats up slowly compared to air.
- So, even on a hot day, swimming pool water often feels cooler.
The same reason explains why oceans help control Earth’s temperature.
3. Cooking Utensils
Cooking utensils are designed using materials with different heat capacities.
- The base is made of metals like aluminum or copper.
- Aluminum: ~900 J/kg°C
- Copper: ~385 J/kg°C
→ These heat up quickly, making cooking faster.
- The handles are made of materials like plastic or wood.
→ These heat slowly, so they stay cooler and safe to touch.
4. Cooling Systems
Water is used in engines and machines because it can absorb a lot of heat.
- It prevents overheating in cars, factories, and power plants.
- Its high heat capacity makes it an effective coolant.
5. Human Body Temperature
The human body contains a lot of water.
- This helps keep body temperature stable around 37°C.
- It prevents sudden temperature changes.
6. Energy Storage
Substances with high heat capacity are used in solar heaters and energy systems.
- They store heat during the day
- Release it slowly later
This improves energy efficiency.
The uses of specific heat capacity are everywhere—from houses and cooking to climate and the human body. It helps control how heat is stored and transferred, making it an important concept in everyday life.
Advanced Perspective: Energy Storage and Efficiency
The high specific heat capacity of water (about 4,186 J/kg°C) makes it one of the most effective substances for storing and managing heat. This property allows water to absorb large amounts of energy with only a small rise in temperature, which is extremely useful in modern energy systems.
1. Thermal Energy Storage Systems
Water is widely used in thermal energy storage (TES) systems because it can store heat for long periods.
- When heated, water absorbs a large amount of energy without becoming excessively hot.
- This stored heat can later be released when needed.
Example:
In buildings, hot water tanks store heat during off-peak hours and release it later for heating. This helps reduce energy costs and improves efficiency.
2. Solar Heating Systems
In solar energy systems, water acts as a natural heat absorber.
- Solar panels transfer heat to water instead of directly heating air.
- Due to its high heat capacity, water can store solar energy during the day and release it at night.
This ensures:
- Continuous heating even after sunset
- Better utilization of solar energy
- Reduced dependence on electricity or fuel
3. Heat Exchangers
Water is commonly used in heat exchangers, which transfer heat between two systems.
- It absorbs heat from hot fluids or gases
- Then carries that heat away efficiently
Used in:
- Power plants
- Air conditioning systems
- Refrigeration units
Because water heats slowly, it can handle large amounts of heat without sudden temperature spikes.
4. Improved Energy Efficiency
The ability of water to resist rapid temperature change helps in maintaining stable systems.
- It reduces energy loss
- Prevents overheating
- Minimizes frequent heating and cooling cycles
This leads to:
- Lower energy consumption
- Longer equipment life
- More stable performance
The high specific heat capacity of water makes it a powerful medium for energy storage and heat transfer. Its ability to absorb, store, and release heat efficiently is why it is used in everything from solar systems to industrial cooling.tions.
Common Misconceptions
- Specific heat vs heat capacity: Not the same
- Higher temperature ≠ higher heat capacity: It’s material-dependent
- Water heats slowly but stores more energy: Often misunderstood
Conclusion
Understanding specific heat capacity is essential for physics, engineering, and environmental science. The specific heat capacity of water stands out due to its high value, making it one of the most important substances for sustaining life and regulating temperature systems.
From the specific heat capacity equation to real-world applications, this concept explains how energy moves and is stored in matter.
Read Next: